Validate that your cleanroom is within specifications.
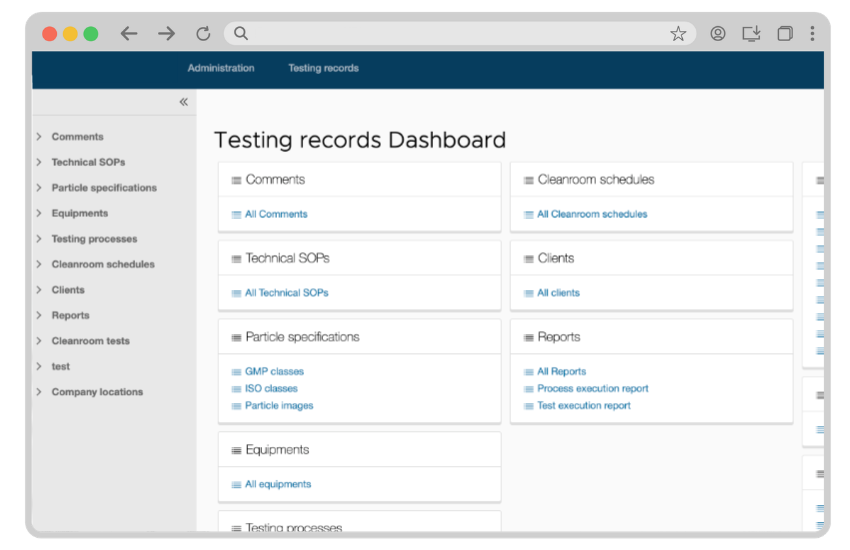
Create testing plans
Create test plans with testing parameters and acceptance criteria.
Provide instructions
Send the test plans to the test engineers along with detailed instructions.

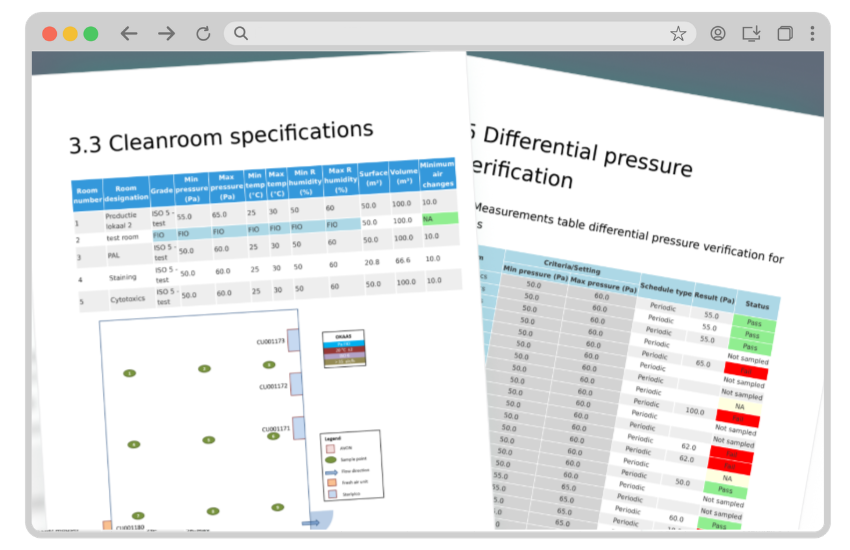
Collect data and generate reports
Approve on- or off-site with electronic signatures. Get reports that prove your cleanroom is within specification.
Digitize and Standardize Your Cleanroom Testing
Solution Overview
Title21 Manufacturing
Download the Title21 Manufacturing Solution Overview to see how Title21 delivers a unified digital manufacturing solution built specifically for cell & gene therapy, CDMOs, and GMP facilities.
Complete the form to download.
See Title21 Manufacturing in Action
Schedule a demo of Title21 Manufacturing and see how teams replace paper, reduce deviations, and gain real-time visibility from batch start to release.



